Molecular Docking of Catechin from Uncaria gambir as a Potential Inhibitor of MurB Enzyme for Antibacterial Drug Development
Original/Research Article
DOI:
https://doi.org/10.33005/jdiversemedres.v3i2.319Keywords:
MurB enzyme, catechin, molecular docking, Pseudomonas aeruginosa, antibacterial agents, peptidoglycan biosynthesisAbstract
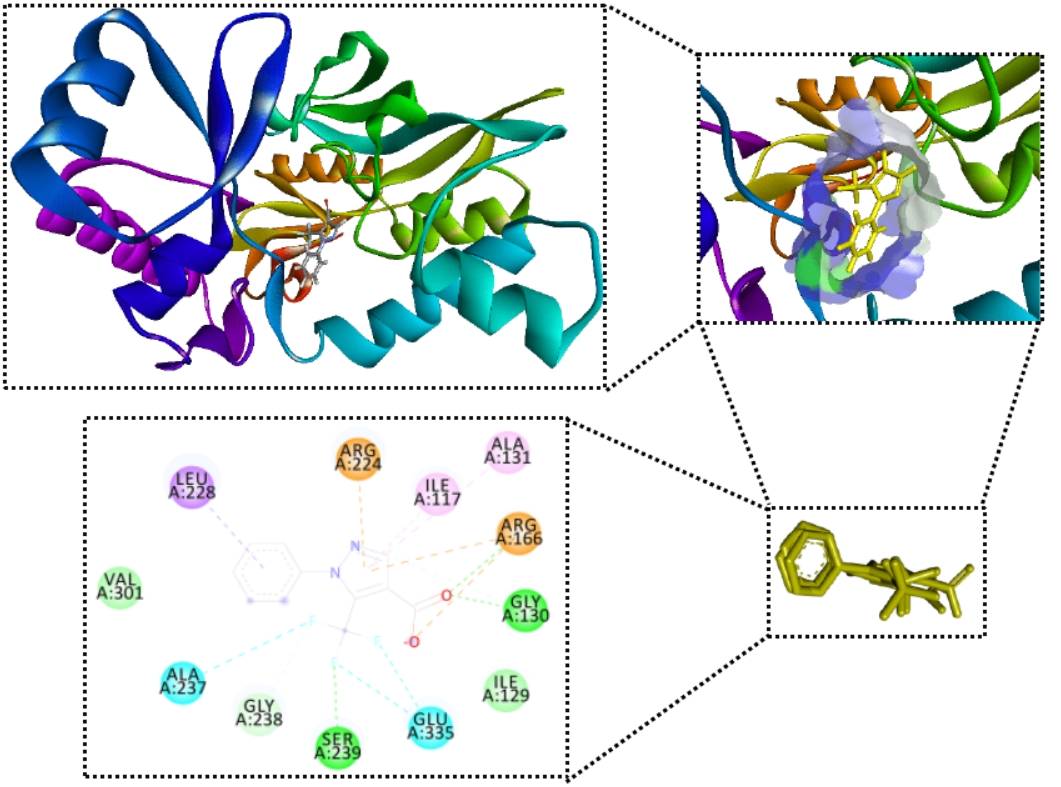
The increasing prevalence of antibiotic-resistant Pseudomonas aeruginosa highlights the need for novel antibacterial agents targeting essential enzymes such as MurB, a key enzyme in peptidoglycan biosynthesis. This study aimed to evaluate the inhibitory potential of catechin against MurB (PDB ID: 7ORZ) using molecular docking. Docking validation was performed through redocking of the co-crystallized ligand, yielding an RMSD of 0.962 Å, confirming the reliability of the method. Catechin exhibited a stronger binding affinity (−7.9 kcal/mol) compared to the reference ligand (−5.2 kcal/mol), with a significantly lower inhibition constant (Ki = 1.63 µM vs. 153 µM), indicating approximately 100-fold higher inhibitory potential. Interaction analysis revealed that catechin forms multiple hydrogen bonds, electrostatic interactions, and hydrophobic contacts with key catalytic residues, including Arg166, Ser239, and Glu335. These interactions suggest that catechin effectively occupies the active site and may interfere with the NADPH-dependent catalytic mechanism of MurB. In conclusion, catechin demonstrates promising potential as a MurB inhibitor and may serve as a lead compound for the development of antibacterial agents against P. aeruginosa. Further experimental validation is required to confirm its biological activity.
Downloads
References
Kováč J, Slobodníková L, Trajčíková E, Rendeková K, Mučaji P, Sychrová A, et al. Therapeutic Potential of Flavonoids and Tannins in Management of Oral Infectious Diseases A Review. Molecules. 2023;28(1):158.
Wu H, Song Q, Wang Y, Manzoor R, Yang X, Li X, et al. Compounds of Plant Origin: The Blue Ocean of Anti-Helicobacter pylori Medicines. Phyther Res. 2025;39(9):4282–307.
Hashim NT, Babiker R, Padmanabhan V, Islam MS, Mohammed R, Priya SP, et al. Polyphenolic compounds in combating MDR periodontal pathogens: current research and future directions. Front Pharmacol. 2025;16.
Pacyga K, Tabiś A, Pacyga P. Medicinal Plants for a Healthy Gut Microbiome: Scientific Insights into Modern Herbal Applications. Int J Mol Sci. 2025;26(22):10875.
Barbosa CD, Rodrigues Santos WC, Da Costa EC, Costa IM, Alvarenga VO, Lacerda IAC. Evaluation of antibacterial activity of black and green tea kombucha. Sci Plena. 2022;18(9).
Singh N. Effectiveness of Green Tea Mouth Rinse over Combination Mouth Rinse in Restoring Salivary pH Post Sugar Exposure in Children. J Sci Res. 2020;64(01):140–3.
Ivyna de Araújo Rêgo R, Guedes Silvestre GF, Ferreira de Melo D, Albino SL, Pimentel MM, Silva Costa Cruz SB, et al. Flavonoids-Rich Plant Extracts Against Helicobacter pylori Infection as Prevention to Gastric Cancer. Front Pharmacol. 2022;13.
Nascimento RC, São José JFB de. Green tea extract: a proposal for fresh vegetable sanitization. Food Sci Technol. 2022;42.
Cheerala VSK, Akhir A, Saxena D, Maitra R, Chopra S, Neelakantan SC. Discovery of benzoxazole-thiazolidinone hybrids as promising antibacterial agents against Staphylococcus aureus and Enterococcus species. RSC Med Chem. 2023;14(9):1712–21.
Kurniawan I, Rohmatika AU. Molecular Docking, QSAR, and Bioactivity Prediction of Uncaria gambir Flavonoids as Antibacterial Agents Targeting MurA Enzyme. Biointerface Res Appl Chem. 2026;16(1):1–22.
Hossain MS, Wazed MA, Asha S, Amin MR, Shimul IM. Dietary Phytochemicals in Health and Disease: Mechanisms, Clinical Evidence, and Applications—A Comprehensive Review. Food Sci Nutr. 2025;13(3).
Kurniawan I. Biokimia Pertanian: Dasar Molekuler Untuk Produktivitas Dan Keberlanjutan (Edisi 1). 1st ed. Wanti Mindari, editor. Lamongan: Ghani Press; 2026. 110 p.
Korol N, Holovko-Kamoshenkova O, Mariychuk R, Slivka M. Insights into bacterial interactions: Comparing fluorine-containing 1,2,4-triazoles to antibiotics using molecular docking and molecular dynamics approaches. Heliyon. 2024;10(17):e37538.
Unver T, Uzuner U, Akcora-Yildiz D, Gurhan I, Arkan C, Ozdemir Z. Multidirectional in silico and in vitro Research for the Pharmaceutical Potential of Fibigia Clypeata (L.) Medik: Phytochemical, Antimicrobial, and Antimyeloma Properties. ChemistryOpen. 2025;14(12).
Kurniawan I, Winarno NS. Unlocking Antibacterial Potential: Thiophene-2-carbaldehyde Modification of Acertannin from African Leaves as MurA Enzyme Inhibitors. J Ners [Internet]. 2025;9(4):7602–12.
Kurniawan I, Ambarsari L, Kurniatin P, Tri Wahyudi S. Novel Compounds Design of Acertannin, Hamamelitannin, and Petunidin-3-Glucoside Typical Compounds of African Leaves (Vernonia amygdalina Del) as Antibacterial Based on QSAR and Molecular Docking. J Jamu Indones. 2024;8:29–38.
Nerdy N, Margata L, Barus BR, Sembiring BM, Ginting S, Bakri TK. Physicochemical , Pharmacokinetics , and Toxicity of South Africa Leaf ( Vernonia amygdalina Delile ) Sesquiterpene Lactone Compounds by In silico: Systematic Review Pharmacy [Internet]. Vol. 12, A multifaceted review journal in the field of pharmacy. academia.edu; 2021. p. 3909–15.
Kurniawan I. Analisis Penambatan Molekuler Turunan Senyawa Tanin Daun Afrika (Vernonia amygdalina Del) terhadap MurA sebagai Antibakteri. Bogor (ID): IPB University; 2021.
Kurniawan I. Modifikasi Struktur Senyawa Tanin Daun Afrika sebagai Antibakteri pada Target MurA dengan Metode QSAR dan Komputasi Dinamika Molekul. Bogor (ID): IPB University; 2022.
Yao H, Gu Y, Zhu D, Tang D, Chen W, Chen Y, et al. Biosynthesis and Application of Catechins and Their Derivatives in Camellia sinensis. Food Sci Nutr. 2025;13(12).
López-Valverde N, López-Valverde A, Montero J, Rodríguez C, Macedo de Sousa B, Aragoneses JM. Antioxidant, anti-inflammatory and antimicrobial activity of natural products in periodontal disease: a comprehensive review. Front Bioeng Biotechnol. 2023;11.
Alibi S, Crespo D, Navas J. Plant-derivatives small molecules with antibacterial activity. Antibiotics. 2021;10(3):1–19.
Ofosu FK, Daliri EBM, Elahi F, Chelliah R, Lee BH, Oh DH. New Insights on the Use of Polyphenols as Natural Preservatives and Their Emerging Safety Concerns. Front Sustain Food Syst. 2020;4.
Kim JW, Kim CY, Kim JH, Jeong JS, Lim JO, Ko JW, et al. Prophylactic catechin-rich green tea extract treatment ameliorates pathogenic enterotoxic Escherichia coli-induced colitis. Pathogens. 2021;10(12):1573.
Mahlake SK, Mnisi CM, Kumanda C, Mthiyane DMN, Montso PK. Green Tea (Camellia sinensis) Products as Alternatives to Antibiotics in Poultry Nutrition: A Review. Antibiotics. 2022;11(5):565.
Yanuarti R, Sartini S, Nainu F. Green tea extract-mediated augmentation of imipenem antibacterial activity against Enterobacter cloacae clinical isolates. Pharmaciana. 2021;11(1):133.
Tocai AC, Rosan CA, Teodorescu AG, Venter AC, Vicas SI. Multifunctional Roles of Medicinal Plants in the Meat Industry: Antioxidant, Antimicrobial, and Color Preservation Perspectives. Plants. 2025;14(17):2737.
Krishna P, Subhan A, Gupta K, Biharee A, Thareja S. Natural Products in the 21st Century: Revolutionizing Therapeutics With Flavonoids as Key Phytometabolites. Phyther Res. 2025;39(9):3833–67.
Mandal MK, Domb AJ. Antimicrobial Activities of Natural Bioactive Polyphenols. Pharmaceutics. 2024;16(6):718.
Verma A, Kumar V, Naik B, Masood Khan J, Singh P, Erik Joakim Saris P, et al. Screening and molecular dynamics simulation of compounds inhibiting MurB enzyme of drug-resistant Mycobacterium tuberculosis: An in-silico approach. Saudi J Biol Sci. 2023;30(8):103730.
Aslam I, Ahmad A, Raja MU, Ahmed S. In vitro compositional and phytochemical analysis of green and black tea. World J Biol Biotechnol. 2023;8(3):19.
Sanad SMH, Ahmed AAM, Mekky AEM. Synthesis, in-vitro and in-silico study of novel thiazoles as potent antibacterial agents and MurB inhibitors. Arch Pharm (Weinheim). 2020;353(4).
Hodyna D, Klipkov A, Kachaeva M, Shulha Y, Gerus I, Metelytsia L, et al. In Silico Design and In Vitro Assessment of Bicyclic Trifluoromethylated Pyrroles as New Antibacterial and Antifungal Agents. Chem Biodivers. 2024;21(8).
Hervin V, Roy V, Agrofoglio LA. Antibiotics and Antibiotic Resistance—Mur Ligases as an Antibacterial Target. Molecules. 2023;28(24):8076.
Ciobanu N, Sucman N, Petrou A, Gorincioi E, Valica V, Uncu L, et al. Biological and Computational Study of the Dual Antimicrobial and Anticancer Potential of 3,4-Dihydropyrimidin-2(1H)-ones and Thiones. ChemistrySelect. 2025;10(26).
Agrahari B, Chaudhary K, Dewan S, Sonker H, Arjun S V., Chandra A, et al. Theragnostic Dual-Action Platform: Ruthenium p-Cymene-Derived Metalloantibiotics With NIR-II Photoacoustic Spectral Signal. Small. 2025;21(26).
Horishny V, Kartsev V, Geronikaki A, Matiychuk V, Petrou A, Glamoclija J, et al. 5-(1H-Indol-3-ylmethylene)-4-oxo-2-thioxothiazolidin-3-yl)alkancarboxylic Acids as Antimicrobial Agents: Synthesis, biological evaluation, and molecular docking studies. Molecules. 2020;25(8):1964.
Kurnia D, Ramadhanty ZF, Ardani AM, Zainuddin A, Dharsono HDA, Satari MH. Bio-mechanism of catechin as pheromone signal inhibitor: Prediction of antibacterial agent action mode by in vitro and in silico study. Molecules. 2021;26(21):6381.
Haque MA, Singh M, Tripathi MK, Ethayathulla AS, Kaur P. Identification of natural small molecule modulators of MurB from Salmonella enterica serovar Typhi Ty2 strain using computational and biophysical approaches. Proteins Struct Funct Bioinforma. 2023;91(3):363–79.
Ribeiro J, Silva V, Igrejas G, Barros L, Heleno SA, Reis FS, et al. Phenolic Compounds from Pyrus communis Residues: Mechanisms of Antibacterial Action and Therapeutic Applications. Antibiotics. 2025;14(3):280.
Sirakanyan S, Kartsev V, Spinelli D, Geronikaki A, Petrou A, Ivanov M, et al. Synthesis and antimicrobial activity of new 2-piperazin-1-yl-N-1,3-thiazol-2-ylacetamides of cyclopenta[c]pyridines and pyrano[3,4-c]pyridines. Arch Pharm (Weinheim). 2021;354(1).
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Journal of Diverse Medical Research : Medicosphere

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
CC Attribution 4.0

